We specialize in FDA approved devices for treatment and diagnosis of neurological ailments with accessible pricing and unmatched support.
Machines built to last, manufactured since 1996 and trusted in 35+ countries.
Fast shipping, efficient training, and human support to get your practice set up in days.
Invite team members and track patients across multiple locations in one dashboard.
Generous warranty + extended coverage at an affordable price for peace of mind.
Encrypted data automatically synced and backed up in real time between locations.
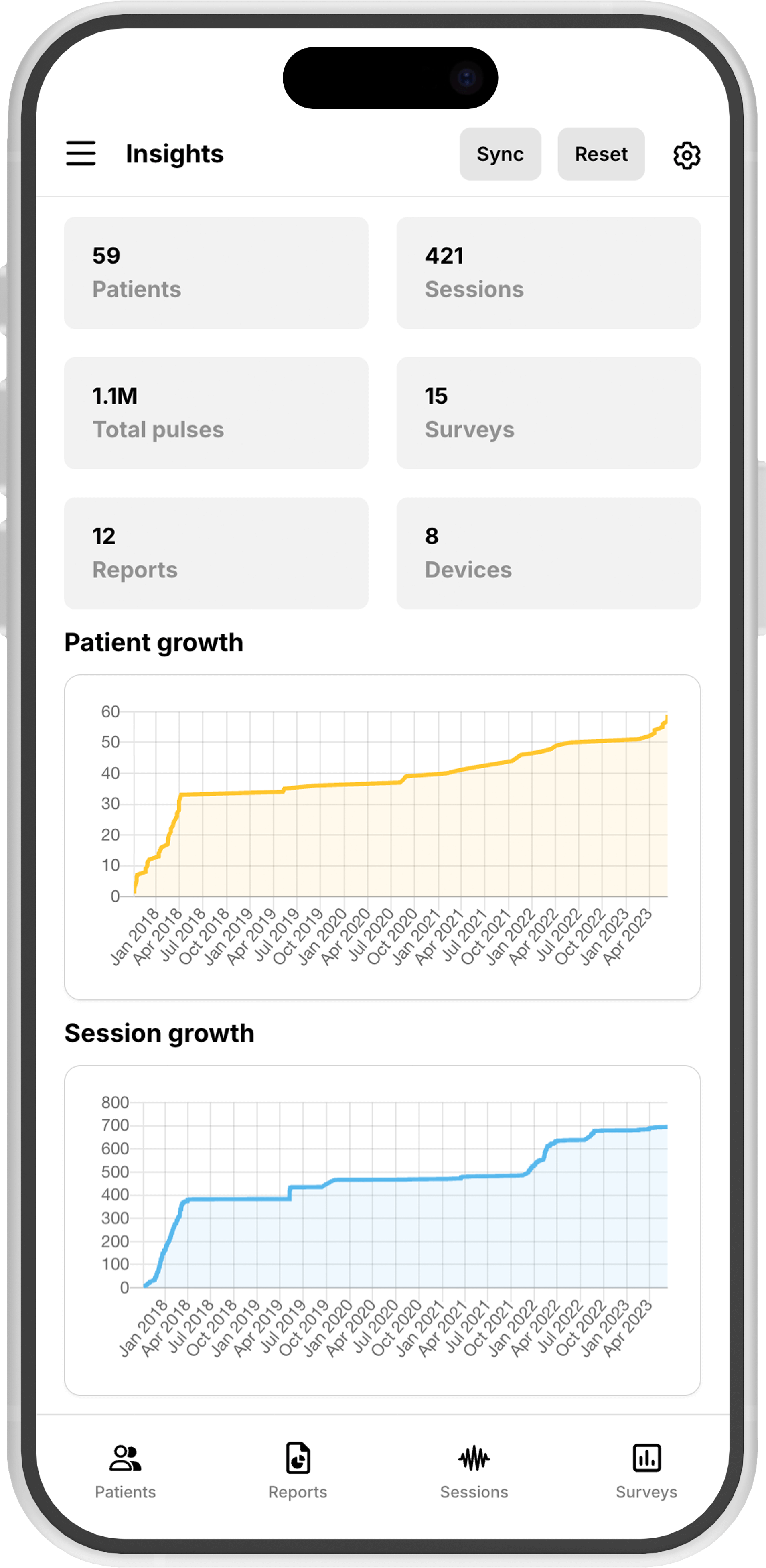
Break down clinic and patient data into useful statistics and downloadable reports.
Patient data in sync
Keep your patient records, treatment plans, and progress tracking synchronized across all practice locations in real-time.






















































Multi-location synchronization
Whether you're managing a single clinic or a network of practices, CloudNeuro keeps all patient data perfectly synchronized. Updates made at one location are instantly reflected everywhere, eliminating data silos and ensuring your team always has access to the latest information.
Seamless care continuity
Patients can move between providers and locations without missing a beat. Treatment histories, session notes, and progress metrics travel with them, enabling any clinician to pick up exactly where another left off and deliver consistent, informed care.
End-to-end encryption
All patient data is encrypted in transit and at rest, ensuring HIPAA compliance and complete data security.




















Bank-level security standards
Every piece of patient information is protected using AES-256 encryption, the same standard trusted by financial institutions worldwide. From the moment data leaves your device to when it's stored in our secure cloud infrastructure, it remains completely unreadable to unauthorized parties.
HIPAA compliant by design
CloudNeuro was built from the ground up with healthcare privacy regulations in mind. Our infrastructure meets all HIPAA technical safeguards, including access controls, audit logs, and automatic session timeouts, giving you peace of mind that your practice remains compliant.


